Veterinary Regulations
The realignment comes at a critical time in the animal drug review program, the agency reports.
USDA has granted Mr. Claus and his reindeer a special permit to enter the U.S., ensuring a seamless journey for the joy they bring each holiday season.
The FDA guidance aims to promote more judicious use of medically important antibiotics in livestock and poultry.
Starting on June 11, over-the-counter livestock antibiotics will no longer be available through traditional retail channels.
On June 11, FDA’s Guidance for Industry #263 brings 91 over-the-counter antimicrobial products from OTC to prescription oversight. Three experts weigh in on why you need to prepare for this change now.
Beginning June 11, 2023, over-the-counter livestock antibiotics will require a veterinary prescription. Producers are encouraged to establish a veterinarian-client-patient relationship now.
The effective date of the decision is Feb. 21, which means that the FDA will again hold veterinarians to the federal requirements for the veterinarian-client-patient relationship beginning on that date
On June 11, 2023, over-the-counter antibiotics will no longer be available through traditional retail channels. Instead, these antibiotics will now require a prescription from a licensed veterinarian.
The American Association of Bovine Practitioners (AABP) recently released a first-of-its-kind document on cattle vaccine guidelines.
Market forces and regulatory actions will drive emphasis on disease prevention and antibiotic stewardship.
Market forces and regulatory actions will drive emphasis on disease prevention and antibiotic stewardship.
The Washington State Department of Agriculture (WSDA) is now offering Washington ranchers a quicker, more cost-effective way to report cattle sales and out-of-state movement of their cattle.
Over the past few years, cannabidiol (CBD) has become wildly popular as a dietary supplement and “natural” treatment for a laundry list of health conditions in humans and animals.
Animal drug compounding can provide tailored treatments for patients or conditions not easily addressed with FDA-approved drugs, but the practice also entails some risks.
Behind the scenes, animal health officials are working on behalf of producers and the livestock industry.
Expanded conditional approval has the potential to incentivize drug development and provide veterinarians with legally marketed new animal drugs.
On August 29, FDA released its first report on inspection and compliance activities.
The Texas Animal Health Commission (TAHC) has proposed a Bovine Viral Diarrhea Virus (BVDV) control program, including designating BVDV as a reportable disease.
In late March, FDA released its draft Guidance for Industry 120, which aims to address questions that emerged since implementation of the current Veterinary Feed Directive rules in 2017.
When the FDA issued its final VFD rules back in 2015, they also released a draft guidance for industry offering answers to anticipated questions regarding application of the rules in common and unusual circumstances.
The United States Department of Agriculture’s (USDA) Animal and Plant Health Inspection Service (APHIS) launched an interactive website that provides livestock producers with important information.
Earlier this year, the FDA provided updates to its Draft Guidance for Industry (GFI) 120, based on stakeholder questions regarding details around the veterinary feed directive (VFD) rules.
Based on comments received regarding a proposed rule, USDA’s Animal and Plant Health Inspection Service (APHIS) announced this week it will partially withdraw the proposal.
When the FDA issued its final veterinary feed directive (VFD) rules back in 2015, they also released a draft guidance for industry (GFI) offering answers to anticipated questions.
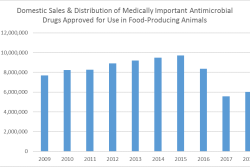
Efforts toward improving stewardship of antibiotics in livestock production have gained momentum worldwide, with more countries regulating antibiotic use and collecting data to document trends.
The USDA’s Animal and Plant Health Inspection Service (APHIS) has published a final rule updating requirements for identifying cattle imported to the United States from Mexico.
Plan includes cost-sharing for electronic ID, birth-to-slaughter traceability.