Beef and dairy producers have adapted to significant changes in antibiotic regulations over the past three years, and that trend will continue. Continued public pressure and regulatory actions will drive ongoing change, with more emphasis on preventive care and veterinarian oversight of antibiotic use.
In September 2019, FDA released draft guidance for industry (GFI) 263, outlining a process for bringing remaining animal drugs containing antimicrobials of medical importance under the oversight of licensed veterinarians. This includes injectable antibiotic products now available over the counter.
The action builds on the GFI 213 and the veterinary feed directive (VFD) rule, which, in January 2017, eliminated performance claims from labels for medically important antibiotics and brought purchase of most medicated feeds under veterinarian oversight.
Also, by September 2020, FDA plans to issue a draft strategy to ensure all medically important antimicrobial drugs used in the feed or drinking water of food-producing animals have a targeted duration of use.
While the industry has made progress in antibiotic stewardship, research shows opportunity for further improvement. The CDC’s 2019 Antibiotic Resistance Threats Report for example, shows an 18% reduction in annual antibiotic-resistance (AR) associated deaths since 2013. Even so, the report notes AR bacteria and fungi cause more than 2.8 million infections and 35,000 deaths in the United States each year.
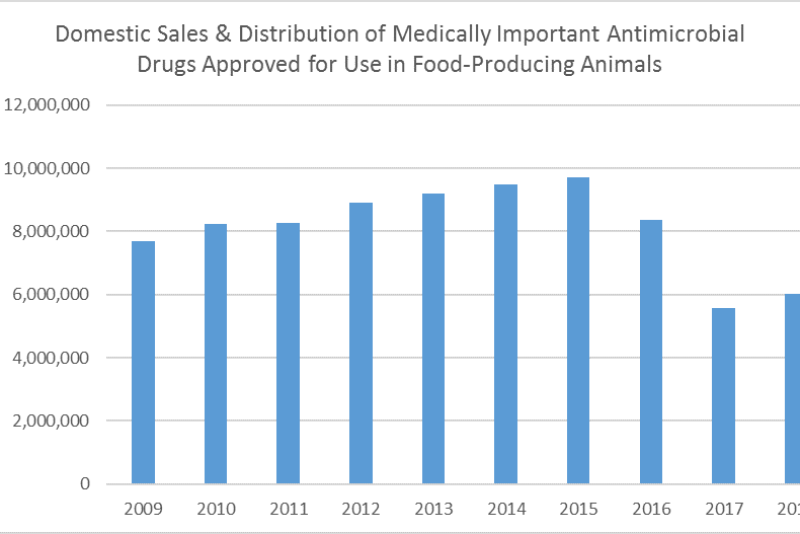
In December, the FDA’s 2018 Summary Report on Antimicrobials Sold or Distributed for Use in Food-Producing Animals showed domestic sales and distribution increased 9% between 2017 and 2018. Without proper context, the year-over-year increase could generate disproportionate reactions. A year earlier, FDA’s summary report showed sales and distribution of those same products declined 33% between 2016 and 2017, and 41% since 2015, which was the peak year for sales and distribution of those products.
While the report does not evaluate reasons behind the year-to-year increase, it seems plausible that 2018 brought a readjustment following a major disruption. Implementation of the VFD rule could have reduced sales below the natural demand level, because of unfamiliarity with the rule and VFD process. Also, sales during 2016 likely reflected effects of some stockpiling ahead of the new rules, making the 2017 decline more dramatic.
FDA notes that although sales data do not necessarily reflect actual antimicrobial use, sales volume observed over time is a valuable indicator of market changes. However, when evaluating progress, we need to consider actual use data, animal demographics, animal health data, and data on resistance.
Several recent reports have shown the livestock industry has made progress toward continuous improvement in antimicrobial stewardship. Nevertheless, activists and consumers will continue to apply pressure for more regulations, and for retail-driven standards beyond those imposed by government. Looking ahead, we’ll need to practice and publicize efforts to improve antibiotic stewardship while protecting animal health, welfare, food safety and producer profitability.
Farm Journal editors have covered this issue extensively, and compiled a comprehensive list of articles to keep you up to date on the latest trends, successes and challenges in antimicrobial stewardship. For links to those articles, visit https://www.bovinevetonline.com/article/antibiotic-resistance-2020-fight-fiction-facts.
Sidebar
Regulatory Timeline
Key FDA Steps toward Greater Antibiotic Stewardship
1996: Together with the USDA and CDC, FDA/CVM established the National Antimicrobial Resistance Monitoring System (NARMS) program.
1997: Prohibited extralabel use of fluoroquinolones and glycopeptides.
2003: Published GFI #152, “Evaluating the Safety of Antimicrobial New Animal Drugs with Regard to Their Microbiological Effects on Bacteria of Human Health Concern.”
2005: Withdrew the approval of fluoroquinolones for use in poultry.
2010: Published first annual summary report on antimicrobials sold or distributed for use in food-producing animals.
2012: Published GFI #209, “The Judicious Use of Medically Important Antimicrobial Drugs in FoodProducing Animals,” establishing a framework for ending production uses of medically important antimicrobials.
2013: Published GFI #213, “New Animal Drugs and New Animal Drug Combination Products Administered in or on Medicated Feed or Drinking Water of Food-Producing Animals.”
2015: Updated the veterinary feed directive (VFD) regulation, governing how veterinarians authorize the use of VFD designated animal drugs in feed and establishing a framework for VFD inspections.
2016: Sought public input on establishing appropriately targeted durations of therapeutic use of medically important antimicrobial drugs in food-producing animals.
2017: Completed implementation of GFI #213, transitioning medically important antimicrobial drugs used in the feed or drinking water from over-thecounter status to VFD or prescription status and eliminating production uses .
2019 FDA releases draft guidance for industry (GFI) #263, outlining a process for voluntarily bringing remaining approved animal drugs containing antimicrobials of human medical importance under the oversight of licensed veterinarians by changing the approved marketing status from over-the-counter (OTC) to prescription (Rx).
2020 (By September) FDA will a draft strategy to ensure that all medically important antimicrobial drugs used in the feed or drinking water of food-producing animals have an appropriately targeted duration of use.