Veterinary Regulations
When state or federal authorities conduct an audit for compliance with veterinary feed directive (VFD) rules, they’ll begin at the feed distributor, working back as needed to veterinarians and producers.
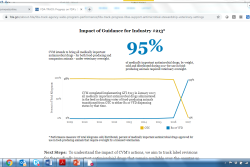
Eighteen months after full implementation of the new VFD rules for medicated feeds, most veterinarians and other stakeholders have become familiar and comfortable with the process and compliance in routine practices.
Research needed to help define duration of use for certain medically important antimicrobial drugs for food animals.
On November 21, 2019, at 1:00-2:30 p.m. EST, the FDA’s Center for Veterinary Medicine will hold a public webinar about the agency’s newly-released draft Guidance for Industry #256.
Kansas State University’s Olathe campus is now offering a premier graduate certificate designed for the regulatory affairs sector of the animal health industry.
Updates to navigation and functionality affect all GVL users.
Performance measures track the progress of the Center of Veterinary Medicine’s (CVM) Five-Year Plan for Supporting Antimicrobial Stewardship in Veterinary Settings.
The United States Animal Health Association (USAHA), the nation’s animal health forum for over a century, is holding its 123rd annual meeting in Providence, Rhode Island, from Oct. 24-30. I
On September 11, 2019, the FDA extended the comment period for its request for public input on zero-day withdrawal periods and zero-day milk discard times for animal drugs.
Like other pathogens and agricultural pests, antibiotic-resistant bacteria do not recognize international borders.
The USDA’s Animal and Plant Health Inspection Service has finalized the Chronic Wasting Disease Herd Certification Program Standards that were originally published on March 29, 2018 for stakeholder feedback.
New data from the National Animal Health Monitoring System (NAHMS) provides a baseline for antibiotic trends in food animal during 2016, prior to implementation of key rule changes.
The Center for Food Security and Public Health at the Iowa State University College of Veterinary Medicine has launched updated versions of two of its online veterinary immunology and principles of vaccinology courses.
The U.S. Department of Agriculture’s (USDA) Animal Plant Health and Inspection Service (APHIS) is updating its scrapie regulations and program standards.
The FDA this week announced an open period for applications for grants to support the development of new animal drugs intended to treat uncommon diseases (minor uses) in major species, or to treat minor species (MUMS).
The AVMA, AAVSB, AAVMC and NAVTA have released a statement of support for licensing requirements. These groups are working to standardize and improve licensure standards for veterinarians and veterinary technicians.
USDA’s Animal and Plant Health Inspection Service (APHIS) recently issued a final rule requiring reporting of all adverse events associated with veterinary biological products.
The Texas Veterinary Medical Association (TVMA) is celebrating National Veterinary Technician Week, which runs from October 16-22, by promoting its new membership for Licensed Veterinary Technicians (LVT).
A good read: “5 Fast Facts About the New FDA Antibiotics Guidelines”. Get the facts on what you need to know!